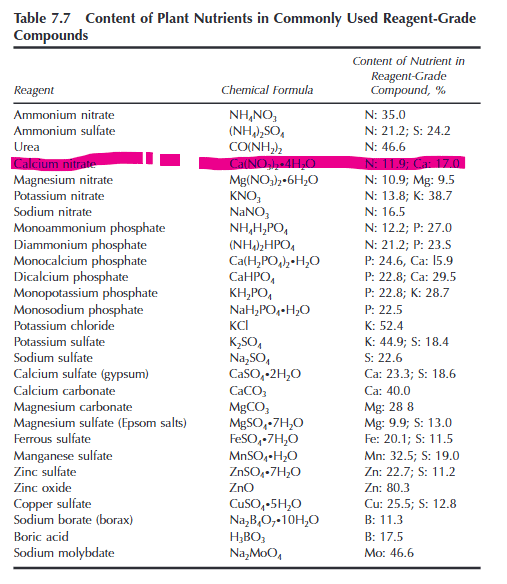
The book says percentage of Nitrogen in Calcium Nitrate is 11.9% but my calculations say 17% 😭
Ca(No3)2 = atoms: 1 Calcium, 2 Nitrogen, 6 oxygen…
now: Molar mass calculations are:
Calcium = 40.08 grams/mol x 1 = 40 grams/mol
Nitrogen = 14.01 grams/mol x 2 = 28.02 grams/mol
Oxygen = 16 grams/mol x 6 = 96 grams/mol
Calcium nitrate total is 164.01 grams/mol..
So percentage of nitrogen in Calcium Nitrate should be (28.02 / 164.10) x 100 = 17.08%
Why does the book say 11.9%?
Update:
Understood it… its not just Ca(No3)2, in the book its Ca(No3)2.4H2O, the extra 4H2O brings the total weight up to 236.18 from 164.01.
So now
(28.02 / 236.18) x 100 = 11.86%
by NnTank

1 Comment
And you’re welcome 😁